

Let us develop and complete an audit ready folder alongside your project. Our offerings include: • A dedicated team of highly skilled and experienced scientists for the duration of the project • Strong customer communication with regular project updates • We can transfer a complete Design History File upon project completion. You have complete ownership • We use development and design control to quality standards within ISO 9001, ISO 13485 and FDA GMP • We are comfortable in designing individual plans for a customised approach to meet your project requirements
The last 5% of any project is the hardest. • In-house high quality reagent and conjugate preparation: gold colloid, silver, platinum, latex (colored, fluorescent and magnetic) • Development of multiple assay formats: antibody or antigen sandwich, competitive, quantitative tests/readers and multi-analyte experience • All forms of sample matrix; whole blood, saliva, stool, urine, semen, food matrix, bacterial cultures, fuel and plant tissue • Off the shelf platforms
We can make complete product on site or transfer to and train your preferred OEM manufacturer in manufacturing your product. • If required for the initial roll-out of your product, we can contract manufacture your lateral flow rapid tests (small scale or large scale) • Mould design and supply of lateral flow device housings

Research, materials discovery and full development under ISO13485
In-house and collaborative design and manufacture of key proteins

Side-by-side modernization of current product

High quality assay components manufactured for 3rd party use
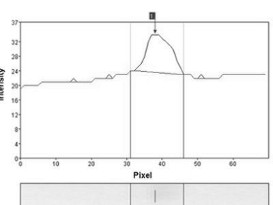
Clear, unambiguous access to your project development phases
Proprietary technology in manufacture and labelling of coloured gold particles

Sensitivity upgrades and dynamic range manipulation

Project management of quality moulding and production instruments
We have a highly focused development and manufacturing company of less than 30 people. Our average experience per head in the medical assay development laboratory is 12 years...

Nick moved from pharmaceutical manufacture into medical diagnostics, and has worked specifically in lateral flow IVD development for 25 years, with companies such as Abbott Diagnostics, Alere, BBI and Vision Biotech.

Julianne is an analytical chemist, who has completed additional advanced post-graduate qualifications in Operations Management and Quality. She has 12 years IVD experience, has developed a significant of new assays, and controls all laboratory manufacturing operations at LFL.

Yumna brings a strong background in Microbiology and QA. She has worked on multiple development projects and taken LFL through several audits with her contributions in QA.

Jenna has developed excellent laboratory and manufacturing credentials. She achieved her B.Sc at Stellenbosch University and is an invaluable member of the development team.

Greg has many years of quality laboratory experience and currently manages the companies operations functions, including managing all non laboratory staff.

Roslyn was originally an analytical chemist, completed an MBA and then moved into Quality Management. She has worked and managed quality sites in some of South Africa's largest companies.

Faeza Williams, Caren da Silva and Cynthia Philander have worked for IVD manufacturing companies for over 15 years each. They bring their supervisory skills to the assembly, stores and packaging functions.

Wendy Parry is our company accountant and maintains our debtors and creditors, orders, all financial queries and our tax compliance. She has an extensive track record in small/medium enterprise accountancy.
If you are looking for a specialist lateral flow development house, with high transparency and cost efficiencies versus our competitors, then let's talk, you may be surprised!
1/2 Greenwich Place
Capricorn Technology Park, Cape Town
+27 (0) 872658241